
ROUGH STONE
"BUBBLES"?
Unlike the perception of many in the Diamond trade, Natural diamonds do not have gas bubbles as is commonly thought, some inclusion looks like the shape and transparency of gas bubbles which can appear hollow. In the extreme temperature and high pressures which cause diamonds to form, there is no possibility of retaining gas in any form.
The only time we might see gas bubbles are if a diamond has been treated with a filling material such as glass.
This treatment is done after a diamond has been cut and polished. Inclusions that look like bubbles are most likely solid foreign minerals inside the diamond. these are a gap in the structure or very small crystals inside the diamond. Occasionally, the mineral wants to expand but the diamond that embraces the mineral does not allow it to expand, penetration of a laser beam or polishing in a sensitive area will allow the imprisoned mineral to erupt into the contact zone as a feather. In fact, there is no need for laser drilling to release the gas, as drilling can result in damage.
The development of new technical equipment has proved conclusively that diamonds do not contain any form of gas.

Feathers
(fracture, "Glatzes")
Feathers are cracks created during diamond formation or damages incurred during the mining of the diamonds. internal fractures or "feathers" can compromise the structural integrity of a diamond.
There are structural differences between different types of feathers and between those that meet the outer surface of the stone and internal feathers that do not touch the surface. the viewing of a Feather between crossed polarizers, by focusing on the Feather peak, indicates a change of behavior from brittle to Elastic behavior.
In addition, a Feather located in the Cleavage direction of the stone is a high potential for damage and must be considered in stone planning, since the cleavage is the inclination of crystalline materials to split along the crystallographic structures.
Inclusions
Diamonds contain the world’s oldest and deepest mineral samples as inclusions. In the extreme temperature and high pressures that creates a diamond to form by layer to layer, it may incorporate small pieces of the surrounding splinters of rocks, a solid foreign crystal or mineral, for instance, carbon atoms located in irregular crystal lattice sites, or vacant lattice sites are intrinsic defects. Impurity atoms are impurity defects. Most defects in diamond are development of comprising intrinsic and impurity defect components.
From practical experience we are able to determine the relative risk the different impurities pose.
When the inclusions have no polaris tension, they hardly effect the durability of the diamond structure.
If the inclusion is surrounded by polaris tension, it means that its growth direction is different from the large stone directions in which it is embedded (a grown-in dislocation) and that affect the integrity of the diamond structure, therefore during the planning and sawing it is important to know which colors showed in the polariscope to avoid, to prevent damage.
Fluoresence
About 35% of all diamonds emit visible glow light when exposed to UV invisible ultraviolet light.
This glow (most common are blue, some will be yellow, orange, green or even white) is called fluorescence and lasts only when the diamond is exposed to the ultraviolet light.
The Fluorescent glow in the diamond does not have any effect on the structure, durability or hardness of the diamond.
Note that exposure to short-wave UV radiation is dangerous for the eyes and should be avoided. Safety glasses should be worn always.

Graining
An "ideal" diamond is composed of carbon atoms packed in a cubic crystal structure. Physical properties, including optical characteristics are uniformly distributed in all directions. Real diamond crystals contain discontinuities in this regular pattern of carbon atoms. When these discontinuities are visible with a 10× loupe or a standard gemological microscope they are referred to as graining. Graining can be caused by primary stresses created during the diamond’s formation or secondary stresses created after a diamond is formed. For example, plastic deformation is a common feature of natural diamonds. Primary stressors include impurities (i.e., other elements, such as nitrogen or boron) that disturb the packing of carbon atoms in the diamond lattice, inclusions of other crystals encapsulated by the diamond during its growth, and grown-on dislocations which are an error in the diamond’s growth pattern due to inclusions. Secondary stressors include plastic deformation which is a permanent disruption in the crystal lattice due to high temperature and heat in the earth’s surface after a diamond is formed.
Polarity
The method is an important tool to discern the critical stress points in a material and is often used for determining stress concentration factors in irregular geometries.
The method is based on the property of birefringence (double refraction), which is exhibited by certain transparent materials.
Diamonds form layer by layer over time.
The number of nitrogen impurities included in each layer varies.
Significant differences between the number of impurities in two close layers create localized stress at the border of the layers.
When a ray of polarized light PASSES through some materials it is transformed along the two principal directions, each of these components experiences different refractive indices. This property is known as birefringence. The difference in the refractive indices causes a relative phase retardation between the two component waves and can be viewed properly in a polarized microscope.
Diamonds exhibit birefringence in areas of internal stress. the magnitude of the refractive indices at each point in the diamond is directly related to the state of stress at that point.
When viewed under a polarized light microscope the phase shift caused by different stress indices is viewed as different color schemes. These color schemes can be interpreted only by an expert who can indicate what stresses and defects caused them.
The scientific interpretation is based on associating the color schemes to internal stress magnitudes and distribution of the risk processing through a set of equations.
However, coloration alone does not indicate risk and therefore a thorough scientific assessment is required to analyze the actual stresses within the stone and the real risk of processing.
Manufacturing a stone without understanding the nature, magnitude (Orders) and distribution of its internal stresses is like marching through a minefield with eyes covered.


TYPES
Diamonds are scientifically divided into four main types The “Type” classification of diamonds is based on the presence or absence of nitrogen and boron and according to the nature of crystallographic defects present and how they affect light absorption, those types can be identified at 100% only by a gemological spectroscope or a FTIR spectrometer (an instrument which measures the amounts of impurities present and absorption of electromagnetic radiation of the diamond by sending an infrared radiation beam through the diamond and measuring how much of it is absorbed):
Type 1 diamonds are subdivided into type 1a and type 1b based on how nitrogen is dispersed throughout the crystal structure since nitrogen is the most common impurity in diamond.
Type 1a:
The majority (97%) of natural diamonds suitable for jewelry are type 1a. They contain two nitrogen atoms adjacent to each other in the structure. (type II diamonds do not contain enough nitrogen to be detected).
Normally their colors can be colorless, near-colourless, cape colour, or brown. They are known as “cape” diamonds because they were originally mined in the Cape area of South Africa.
Type 1b:
Not many diamonds were found to be Type 1b, in these diamonds, single nitrogen atoms that have replaced carbon atoms in the structure are isolated from one another, they do not occur in adjacent structural positions, these diamonds are almost always brown, bright yellow, or orange and are extremely rare, from the Zimmi mining area ( Mano River at Liberia) in West Africa, their durability and hardness might be weaker in comparison to other diamonds, especially when inclusions are presents.
Type 2:
Type 2 diamonds almost always show a cross-hatched “tatami” strain pattern under a microscope between crossed-polarized lenses.
Type 2a:
Very rare in nature (1.7%), type II diamonds do not contain enough nitrogen or boron impurities to be detected by IR or UV absorption measurements. they are often having no solid inclusions.
They appear in nature in these following colors: Colorless, light brown, light yellow, light pink or gray, and have the highest thermal conductivity.
Type 2a has a stronger structure than other three types.
Type 2b:
Make up about 0.1% of all-natural diamonds These diamonds, like Type 2a, contain almost no nitrogen impurities that can't be easily detected by IR or UV absorption measurements,
But they contain boron impurities that are thought to be isolated single atoms that replace carbon in the diamond structure.
The trace element boron is responsible for most diamonds that are blue or grayish blue in color. The absorption spectrum of boron causes these gems to absorb red, orange, and yellow light, lending Type 2b diamonds a light blue or grey color,
Type IIb rarely exhibit tinge that is outside the blue-gray-violet range.
fancy color
Impurities such as nitrogen and plastic deformation of the diamond structure are the causes of color in diamonds.
Pink diamonds:
Pink, Purple and red colour is due to plastic deformation of the diamond structure which can result in displacement of the carbon atoms along glide planes. Brown diamonds typically contain nitrogen impurities and graining associated with deformation of the diamond structure. pink diamonds are most likely to have inclusions.
science has not yet been able to definitively identify why this crystal deformation can lead to specific coloration.
Plastic deformation does not consider a significant risk.
Blue diamonds:
Blue diamonds are extremely rare. They generally have a slight tinge of gray; the intensity of blue color is correlated to the presence of active boron impurities and usually classified as a type 2b diamonds.
Chameleon diamonds change colour, from grey-green to yellow, with a change in temperature, thermochromic response or when exposed to light. These diamonds contain high concentrations of hydrogen along with some nitrogen and nickel (Fritsch et al., 2007a).
Green diamonds:
Green diamonds are not restricted to any diamond type, receiving their colour from exposure to ionizing radiation (nickel-related \GR1 center) which causes them to reflect typically light in tone and low in saturation. Their colour often appears dull, with a green or brown tinge, get their colour by displacing the carbon atoms from their usual places in the crystal structure. This can happen naturally when diamond located near radioactive minerals.
The color- tone of most of green diamonds is found on the outer surface, and rarely through the entire stone.
To enhance the color, before polishing the rough, several steps must be taken in order to retain the rough green color at the polished diamond.
Black diamonds:
Black diamonds are usually colored by cloud inclusions, graphite magnetite, hematite, iron or other black mineral inclusions located in the stone. (common features for this 1a type).
Undetermined:
The GIA is not Shure of the origin of the color which reflects to whether the stone is natural, or color treated.
Not Applicable:
The GIA normally grades the color distribution of “Even” or “Uneven” stones and hence grades the stone Applicable or “Not Applicable” grade.
Most of all colored diamonds graded by the GIA fall into a “Fancy” grade description,
but some stones are too weak, Light, Very Light and Faint: these grades will receive always the “Not Applicable” grade.
HPHT
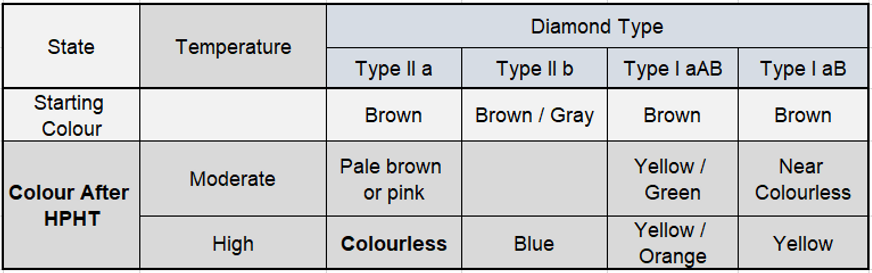
HPHT treatment means High pressure and heating to high temperatures of temperature 1700-2500 C°. under pressure 6 GPa.
HPHT treatments are considered stable and permanent.
In the year 1960, researchers from the Soviet Union reported Experiments in HPHT treatment that it changes the state of nitrogen impurities in the diamonds.
The changes are from single nitrogen (1b) to nitrogen aggregates (1a), and from 1a to 1b. by changing the nitrogen optical defects deformed regions in these stones.
At that time, HPHT treatment could not change brown diamonds to be colorless.
only in 1999 General Electric and Lazare Kaplan International publicized the commercial use of the HPHT process to remove color from type 2a diamonds.
This development aroused turmoil in the diamond industry because of the possibilities created by using this process.
Later over the years in order to detect any possibility of treatment, the GIA started to use high Tec equipment such as PL spectroscopy at liquid nitrogen temperature with several laser excitations, and FTIR (Fourier Transform infrared) mapping techniques. Raman spectroscopy soon became the most common technique used to measure these elastic effects.
certain treatments change type 2 diamonds to pink or blue colours
When a type 1brown stone is subjected to a similar treatment, the presence of nitrogen impurities causes it to change to different shades of yellow.
Extensive knowledge in the preparation process of the stone must be done before a stone can be handed over for this process, it is also important that after the stone returns from the process, it needs to be rechecked in order to protect the stone from being Damaged.
Manufacturing
The information above is to assist in the prevention of damage during manufacturing examining factors which are relevant, as the risk of damage during manufacturing is not necessarily caused by bad luck but due to lack of knowledge and experience.
A rough diamond that is not potentially dangerous should not be damaged, if the damage is caused, then the cause could be due to the equipment, balance, thickness, and speed of the wheel.
To significantly reduce the possibility of damage during manufacturing, several steps must be taken, some of which are complicated, difficult to enforce and require professional knowledge.
Cutting tool:
The tang is the tool which holds the diamond by the polisher, for placing it on the rotating polishing wheel.
the tang consists of two main parts - the arm, with the fixed legs on one side that can be adjusted with screws, and the head \ dop is formed that holds the diamond by holding pliers or hoop.
The top of the tang can be tilted by a dialer to any desired angle.
The tang is used in an environment saturated with diamond dust, which causes deformation, a thermo-mechanical phenomenon that causes deformation, mechanical wear, and freedom (spike\shpill) in the instrument's motion paths in a way that is not noticeable to an inexperienced eye.
A proper examination of the tang locates defects that damage the stability of the tang which leads to damage.
The polishing wheel:
Made from cast iron disc rotates by direct connection to the axis of an electric motor.
The upper part of the wheel is smeared with diamond powder to polish the diamonds.
The diameter of the wheel is about 35 cm, about 2.5 cm in length, and it rotates at 3,000 - 5,000 rpm.
The speed:
The speed rpm and the type of device it connects to the engine have an important effect in preventing damage. One of the essential requirements is that the wheel is balanced to limit vibration.
Most of the poishers say they can feel the vibration and replace a shaking wheel, but unfortunately, only a sophisticated and advanced digital system for wheel balancing can detect minor tremors, and only using such a system allows accurate balancing of the wheel.
The thickness:
The thickness of the polishing wheel also throws damage, and once again an example from the vehicle worlds, when the braking plates in the vehicle front wheels wear out and become thin, the effect refract during braking as a tremor and squeak.
The stone grip in the dop - the routine operation of the polishing work and the polishing concept - is very important.
The stone should be glued to the pot at all stages of polishing. adhesion is necessary, the only disadvantage of adhesion is the hassle and inconvenience that accompanies this action and therefore this stage is not adopted by many polishers.
The way of work:
Laying the tang on the wheel calmly, without many movements using moderate weight, choosing the pot and the appropriate nails and controlled cooling of the stone is part of the skill of the polisher.
Our duty is to convey that the above processes and criteria are the cornerstones of diamond manufacturing. polishers who meet these criteria will be compensated accordingly, since only training in combination with appropriate tang compensation will yield the results of damage prevention during diamond manufacturing.
acknowledgments
Oren Pedatzur PhD Weizmann Institute of Science.
Lourie Ronald for his Technical assistance and proofreading.
Carl Chilstrom, GG, MBA, of the GIA Laboratory.
Magnezi Liel.
REFERENCES:
Thomas M. Moses, Ilene M. Reinitz, Mary L. Johnson, John M. King, and James E. Shigley.
John M. King, and Wuyi Wang.
Jana Fridrichová, Peter Bačík, Radek Škoda & Peter Antal.
Christopher M. Breeding and James E. Shigley